Point-of-Care AM Propels Patient Care
3D printing can unleash a new generation of patient-specific medical devices, but regulatory and skills challenges persist.

The WeMed SKOP, a biomimicry designed, connected stethoscope, shows what’s possible when you unleash additive manufacturing on medical devices for PoC telehealth applications. Image courtesy of Nexa3D.
Latest News
August 4, 2022
The Hospital for Special Surgery (HSS) has long been a mecca for patients stymied by failed joint replacements or who have unique anatomical issues that prevent use of off-the-shelf orthopedic implants. Today, the leading academic medical center focused on musculoskeletal health is also a pioneer for point-of-care (PoC) 3D printing, and is creating bespoke solutions that tackle the most complex orthopedic cases.
In partnership with orthopedics manufacturer LimaCorporate S.p.A., HSS has launched the ProMade PoC Center, a U.S. Food and Drug Administration-regulated commercial facility at the hospital’s main New York City campus where surgeons and ProMade engineers can collaborate on-site.
By establishing a custom 3D printing center, HSS took a major step enhancing collaboration between the engineering and surgical teams while significantly reducing the lead time between designing a custom medical device and deploying it in an actual patient surgery, according to Douglas Leach, vice president for device innovation at HSS.
“It has enabled us to accelerate time to care for our patients, and the secondary benefit is that physicians get to work directly with the manufacturer,” Leach says. “Being able to sit down with everyone involved in the design makes it a much more productive process compared to dealing with distance and time challenges.”
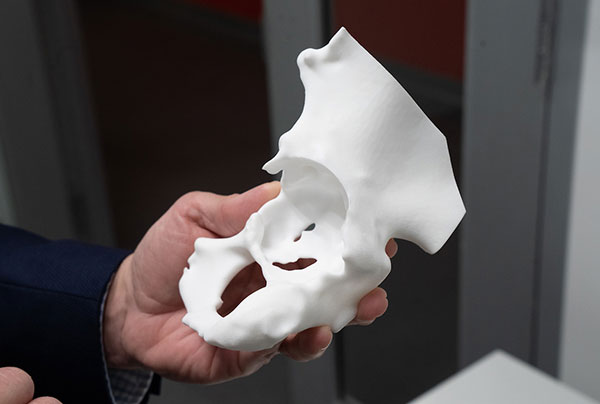
HSS, the Mayo Clinic and the Veterans Health Administration (VHA) are among the healthcare organizations championing PoC 3D printing. With improvements in the ability to convert two-dimensional data from magnetic resonance imaging and computed tomography scans into a format compatible with 3D modeling combined with materials advances and lower cost, more accessible 3D printer technologies are fueling rising interest in creating personalized medical devices to drive better patient outcomes.
“If you look at medicine in general, there’s a trend towards precision medicine that’s well accepted, from gene therapy for addressing disease to therapeutics mixed for someone’s individual make-up,” says Gary Turner, managing director of AM for Ricoh USA, which offers the Ricoh 3D for Healthcare, a PoC 3D printed anatomic modeling solution.
“There is recognition that no two people are the same, so treating them the same is not an effective way of healthcare. That’s why we are seeing the focus in the AM space as part of a broader trend towards personalized and precision medicine.”
Fallout from the global pandemic also put a spotlight on 3D printing’s current and future potential for improving healthcare, from its initial use for outputting much-needed personal protective equipment (PPE) such as masks, face shields and nasal swabs to fast-tracking new devices for telehealth visits.
For example, HP, 3D Systems and Massivit 3D Printing Technologies were among the 3D printer manufacturers donating products and manufacturing resources for producing much-needed PPE during the height of the pandemic.
In an entirely different example, a collaboration between 3D printer maker Nexa3D, polymers developer Henkel and WeMed, resulted in the SKOP connected stethoscope, which leverages biomimicry concepts and cochlear technologies to enable high-level acoustic qualities necessary for remote medical diagnoses. The WeMed device was 3D printed using Nexa3D NXE400 printers to achieve high-volume precision for output at scale and in response to the global call for democratized, affordable teleconsulting healthcare solutions during and after the pandemic, officials say.
“COVID opened everyone’s eyes to the importance of AM as part of a digital health strategy,” says Scott Drikakis, healthcare segment leader for the Americas at Stratasys. “Now the question is: what type of infrastructure does a PoC facility need in order to do this responsibly and at scale? That’s where the conversations are now.”
Device Applications Take Shape
The first wave of PoC 3D-printed medical devices are fairly well established. Hospitals and care facilities have leveraged myriad AM technologies and materials these last few years to output customized surgical guides, custom dental appliances and anatomically correct organ models that can help surgeons and caregivers better prepare for complex procedures and inform patients of what to expect from treatments. With the advent of new biomaterials as well as less expensive metal AM technologies, researchers and some healthcare facilities are expanding practices with efforts to create customized joint and cranio-maxillofacial implants at PoC.
Despite progress, it’s still early on in the PoC AM revolution. Due to high costs and lack of knowledge about what’s available, most small- to mid-sized hospitals still don’t have access to production 3D printers that can output bespoke, medical-grade devices at scale. There are also significant regulatory requirements governing the operations of a medical device-grade manufacturing facility, and most hospitals currently lack the on-staff talent, domain expertise and process capabilities necessary for compliance.
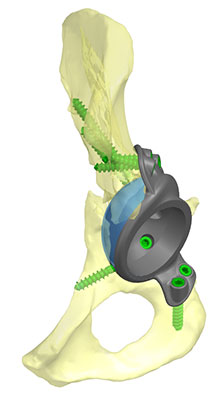
Still, a handful of pioneers are leading the way, including HSS, which has already turned out 3D printed implants for hip and shoulder procedures that precisely match requirements of individual patients. Over time, the group anticipates expanding PoC AM capabilities beyond large joint implants to smaller hand and wrist implants along with plates and screws that can be customized to address the needs of trauma patients in a more timely fashion.
“I don’t think we’ll ever get to PoC AM for regular hip and knee replacements because it doesn’t make sense from an economic perspective,” Leach says. “But for complex cases, that’s where the huge advantage will be.”
The VHA isn’t yet using PoC 3D printing to create bespoke implants, but it does plan to move in that direction; currently, it is actively using AM to create patient-specific organ models and surgical cutting guides at PoC, says Dr. Beth Ripley, deputy chief of the office of healthcare innovation and learning at the U.S. Department of Veteran Affairs.
Ripley also sees opportunity for PoC AM to make safety devices and other healthcare-related tools that might not make economic sense for a third party to manufacture. For example, she cited a recently designed device that serves as a thermal cover for VHA patients using home oxygen equipment to prevent risk of flammability.
“We could go to a third party to make this, but it’s probably not worth it because they wouldn’t make a profit,” she explains. “But it’s essential for us in terms of patient safety and comfort.”
A Challenging Road Ahead
To beef up capabilities in this area, VHA is collaborating with 3D Systems to establish U.S. Food and Drug Administration (FDA) compliant medical device manufacturing processes, the goal being to streamline its supply chain and to promote innovative, personalized care to veteran patients.
As part of the collaboration, 3D Systems is helping VHA develop new workloads and create a quality management system that will ensure full FDA compliance for any on-site 3D-printed devices. 3D Systems is also supplying printers, materials and workflow software to facilitate the VHA’s PoC AM charter along with documentation and training.
Regulatory compliance is one of the biggest obstacles to hospitals and care centers transitioning to a PoC AM model. While doctors have been able to develop one-off custom solutions to complex medical problems without onerous regulatory oversight, that all changes once there is persistent manufacturing of medical-grade devices. FDA policies and procedures become stricter with increased patient risk, especially for Class III implantable devices. The extensive paperwork related to filing a 510(k) to the FDA can be daunting for those not accustomed to the process and especially for hospitals that likely don’t have the right tools and quality management systems in place to support FDA compliance.
“How does a hospital set up a manufacturing capacity, how does it control processes, and reduce and control risk of injury to patients?” asks Ben Johnson, vice president, portfolio and regulatory, for 3D Systems. “Those are things a medical device manufacturer knows really well, but hospitals don’t have the same frameworks and infrastructure in place.”
Quality control systems are key to tackling the regulatory landscape, which is why VHA and other early PoC AM adopters are leaning into collaborating with partners. Ricoh and Stratasys, for example, are partnering to deliver RICOH 3D for Healthcare, an integrated, end-to-end solution for delivering patient-specific 3D anatomic models at the PoC. The system combines Stratasys printers, integration with IBM Watson Health and Ricoh’s quality management system (QMS) and manufacturing best practices. Customers can opt for a PoC option, managed by Ricoh staff, or an on-demand service where they order anatomic models that are printed off-site and delivered.
EOS Formiga P110 3D polymer laser printer is used for producing orthopedic bone models and single-use instrumentation. Image courtesy of HSS.
Being able to tap into PoC AM managed services and fully designed QMS capabilities is critical, notes Ricoh’s Turner.
“Creating a QMS is difficult, complex and expensive—a medical device manufacturer or big hospital like Mayo Clinic can do that, but rank and file hospitals can’t,” he explains.
Hospitals are also not in the business of manufacturing so most are lacking the appropriate staff required to run these PoC AM sites, including process and controls engineers. In addition, a PoC AM center requires experts familiar with digital 3D modeling as well as those versed in running and optimizing complex 3D printers, including proficiency with part orientation and materials knowledge.
“It requires mechanical and materials engineering know-how at a post-degree level and you have to have enough demand to justify hiring those types of people in the workplace,” notes Phil Reeves, director of 3D printing consultancy Reeves Insight.
Training and education on what’s possible is another hurdle—though the education portion is being addressed through university and graduate-level classes like the one taught by Dr. Steven Kurtz, research professor in the Biomedical Engineering Science and Health Systems department at Drexel University.
Kurtz’s seminar, “Point-of-Care Design and Additive Manufacturing of Polyaryletherketone (PAEK) Implants,” provides instruction in how biomedical engineers can work side by side with radiologists and surgeons to produce patient-specific implants, including developing such skills as medical image segmentation, device design and verification and validation.
“The most basic challenge is training and education for the staff at the hospital that’s going to be doing this,” Kurtz says.
Another major hurdle is ironing out the billing and insurance coverage for these devices, especially since they are manufactured as one-offs, according to Mark Burnham, director of policy for Additive Manufacturing Coalition, a national membership organization that aims to be the voice of AM with stakeholders in federal agencies and Congress.
“When you’re talking about customized health care, that’s a paradigm shift,” he says. “There’s not an understanding of systems or the scales in place so there needs to be a really big push.”
It’s still the early days for PoC AM, and these issues remain in the forefront. Hospitals and care centers interested in jumping are certain to take different approaches, depending on the size and scope of their budget and resources. While there is no one-size-fits-all solution, it’s clear whatever model is chosen must place an emphasis on collaboration and co-creation.
“We have hybrid teams of 3D Systems and VA staff working together to create solutions so everything is built through the VA and vet lens,” explains VHA’s Ripley. “The goal is not for 3D Systems to make something off in a corner, but rather work with us to understand and give us the tools, know-how and expertise that allows us to carry forward on our own.”
More 3D Systems Coverage
Subscribe to our FREE magazine, FREE email newsletters or both!
Latest News
About the Author
Beth Stackpole is a contributing editor to Digital Engineering. Send e-mail about this article to DE-Editors@digitaleng.news.
Follow DE