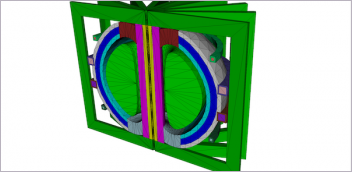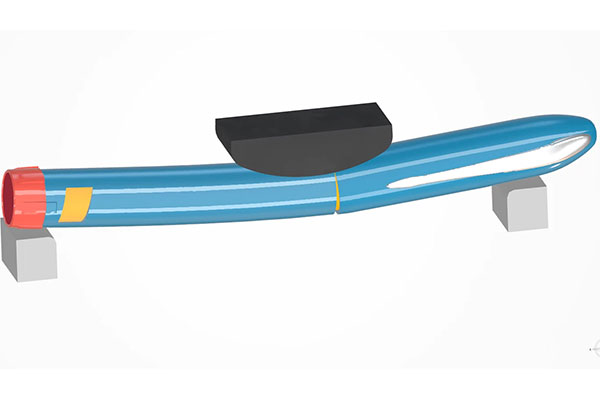
As more medical devices are becoming smart and connected by embedding electronics for scheduling, monitoring, measuring and reporting, electronic performance and regulatory requirements must also be considered. Image courtesy of Dassault Systèmes.
Latest News
August 1, 2022
As engineering design becomes more sophisticated with advances in software, simulation and computing hardware, the products engineers become more innovative. For medical device design, this is so true, despite challenges that prevail for biomedical engineers and other device designers.
As medical device design gains design and engineering resources, so does the wide and deep field of medicine. Once again, engineering—in its totality—provides solutions to problems that previously remained unsolved.
Surmountable Challenges
Medical devices are becoming quite advanced with numerous tools used to expedite their design, but there remain many challenges to bringing such devices to market.
The medical technology (medtech) industry is going through unprecedented disruption, coupled with new demands brought forth by the COVID-19 pandemic, enabling new innovations but also associated challenges, notes Aruna Narayanan, principal software architect for digital at GE Healthcare.
“Beyond that, not all current challenges are completely new; some are so called ‘teething trouble’ due to the adoption of emerging new technologies like [artificial intelligence (AI)] and cloud in the medtech industry,” she says.
Notable challenges, she points out, include disconnected systems. “In many cases, hospitals have disconnected systems from multiple vendors that don’t talk to each other, and it is incumbent upon hospital IT departments to integrate them, connect them together and recruit IT resources to develop and maintain any custom solutions that are built,” she says.
But she also notes there is a data challenge. “With medtech devices producing a huge volume of data, selecting the correct data to identify and solve a specific problem is a challenge,” she adds. “Building AI solutions requires the right volume, variety and veracity of data, and quality of data plays a significant role.
Is it easy to search and retrieve the right data? How do you handle the large volume of data that is generated by the device itself? Does the device have enough compute power and capacity to handle the growing AI applications? Or will there be a need to upgrade or replace a device configuration for the growing computation needs? Should the application run in different environments with a variety of computer resources?
Designers also face challenges with application updates and feedback loops. “With new innovations,” she notes, “how [do you] get feedback that the solution works? How do we enable automated monitoring, get feedback, act on the feedback and deploy a better solution with a faster turnaround?”
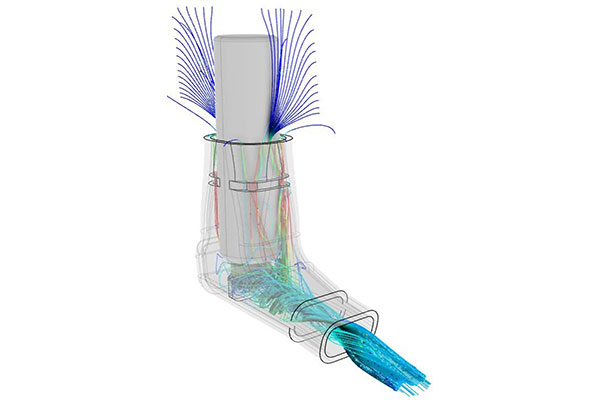
Karl D’Souza is the SIMULIA life sciences and healthcare industry process director at Dassault Systèmes in the greater Boston area. D’Souza says the medical device industry is motivated to deliver sustainable, innovative solutions driven by new biological therapies, wearable devices, patient comfort and the potential to transform patient care. But the variety of needed medical devices can pose issues.
“Scientific and mechanical engineering developments are enabling scientists, engineers and designers to harness our macro and micro worlds concurrently. Advances in material engineering are enabling the selection of specific molecules, biologics, materials and delivery devices to improve patient care and therapeutic outcomes,” he explains. “Key challenges facing medical device makers today include improving the range and value of novel medical devices to satisfy unmet patient needs, achieving sustainability goals and reducing a product’s carbon footprint, and advancing the patient experience with easy-to-use self-care options and positive treatment outcomes.”
D’Souza says these challenges can be addressed with the help of new virtual human models combined with integrated design and simulation technologies.
“The combination enables the use of virtual twins to accelerate the development of smart, connected devices that can also be manufactured with minimal waste and are easy to use, safe and reliable. Additionally, device manufacturers can employ augmented, virtual reality and 3D printing not only during the product design phase, but also for medical training, patient education and surgical planning.”
More Tools in the Engineer’s Toolbox
As engineering software and design technology advance, biomedical engineers have many tools and resources they can turn to. Michael Speier is a senior mechanical engineer at Eitan Medical. Software and development products must be coordinated with many other factors, he says.

“Medical device engineers may use tools such as 3D design software to develop products, but the ability to work in tandem with other teams—regulatory, human factors, design experts and production teams—from the get-go, is just as important,” says Speier. “During the development process, each milestone should be signed off on by all relevant departments to ensure the entire team is onboard with every phase of the design. Another key step is for design experts to help with design tolerance analysis, mold flow analysis, finite element analysis to ensure that the final product will work the ‘first time’ with few to no iterations.”
The use of rapid prototyping technologies, such as 3D printing and CNC parts, to prototype a drug delivery system meant design verification time was significantly reduced by performing an extensive range of pre-verification tests and simulations with quickly produced parts, he explains.
Speier points out that there are many potential benefits of new and developing tools to help present detail in designs. The use of many new and sophisticated tools and expanded resources means products may reach the market within a reduced timeframe.
“Rapid prototyping, preliminary testing and simulations significantly reduce the risk of project failure by allowing early benchtop testing in advance of molding and mass production. Rapid prototyping is not only a great tool throughout product development but can also allow for early feasibility assessment and patient preference studies done by potential partners and customers,” he says.
As patients demand products with greater complexity but lower costs, medical device makers must embrace modern technologies to stay competitive.
“Innovators that provide a compelling and intuitive user experience have a competitive advantage. However, today’s device design process is at a breaking point,” D’Souza says. “No longer can device companies easily meet tight performance, quality, time to market and budget constraints using legacy processes and tools.”
D’Souza adds that despite widespread adoption, 3D modeling and simulation have remained significantly distinct fields, characterized by distinct sets of tools and IT environments customized for designers, pre-processing engineers and simulation analysts working independently of each other.
“Interaction between these groups often requires non-value-added tasks such as model translation between the tools, version control management and other specialized interventions that prolong the development cycle. Moreover, as these functional teams work in silos, there is little chance of meaningful collaboration between them or with other stakeholders across the enterprise,” says D’Souza. “Furthermore, simulation expertise is mostly limited to a few experts, making it difficult to scale and limiting its practical value to the organization.”
To address these challenges, engineers are turning to automation and data science tools that help with test generation and validation. This increases the ability to collect real-world evidence and benefits areas such as digital clinical trials, explains Sheetal Chawla VP, partner, head of life sciences at Capgemini Americas.
“Augmented reality and virtual reality tools are being utilized for physician training, which has led to improvements in areas such as therapy adoption. Nanotechnologies and photonics are being utilized for developing wearable medical devices while enabling a better patient and wellness experience. A significant increase in the use of data and analytics has led to advancements in quality control and the discovery of new therapies,” says Chawla.

Shaping the Future with Medical Device Design
Dan Hermansen is the director of medical illustration at Osso VR, based in Salt Lake City, UT. Hermansen says that as surgical technology and techniques advance, the number and complexity of these procedures are ballooning while the time within which surgeons must learn them is decreasing.
“The learning curve for modern surgery involving technologies such as robotics and patient specific implants is estimated to be between 50 to 100 cases,” says Hermansen. “Seeing a procedure once and then doing it intuitively seems like it would put patients at risk, and it does.”
He says complication rates related to a new technique can be up to 300% higher than normal. The medical world is on track to continue progressing to meet the needs of patients, but medical education needs a reliable repeatable solution to address the needs of healthcare providers worldwide.
VR surgical training and assessment, according to Hermansen, enables healthcare professionals to gain surgical workflow knowledge and demonstrate that they have mastered the surgical skills necessary before ever performing a technique on a patient.
“Similar to modern pilots, who must demonstrate their aptitude within a simulator before taking the lives of passengers into their hands, so too should surgeons be enabled with technology that will ensure the safe practice of medicine within operating rooms everywhere,” Hermansen adds.
“Whatever the technology or tools used, it is important for the designers of the MedTech solutions to remember the noble cause they are part of the minute they find a job in the medtech industry,” says Narayanan of GE Healthcare. “We are contributing to saving or improving lives in moments that matter. It is important this responsibility drives our everyday decisions, beyond creating solutions and efficiencies.”
More Dassault Systemes Coverage

Subscribe to our FREE magazine, FREE email newsletters or both!
Latest News
About the Author
Jim Romeo is a freelance writer based in Chesapeake, VA. Send e-mail about this article to DE-Editors@digitaleng.news.
Follow DE