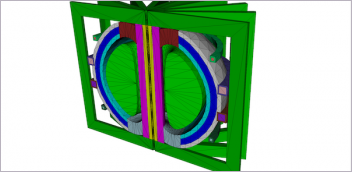
Siemens is making its Additive Manufacturing Network available to help create medical components. Other 3D printer companies, universities, and technology providers are contributing knowledge, materials, and manufacturing capacity to address healthcare equipment shortages. Image courtesy of Siemens.
Latest News
April 10, 2020
The current COVID-19 crisis has brought to light a critical problem in the medical supply chain in the U.S. With many supplies such as personal protective equipment (PPE) coming from overseas, healthcare providers face shortages of masks, gloves, face shields, and other supplies. In addition, the current supply of ventilators was woefully low even before the pandemic.
With its rapid and distributed production capabilities, additive manufacturing technology was literally built for such a moment. Printer manufacturers, technology companies, and engineers are stepping up to help fill the gap in the medical supply chain—and more help is needed.
Universities (and some high schools, and individuals working in livings and garages) across the country have converted their engineering departments and 3D print labs into make-shift factories for churning out PPE. On-demand manufacturing and prototyping operations, as well as manufacturers of other types of products, have converted all or part of their capacity to address the shortages. Hobbyists and makerspaces are pitching with 3D-printed face shields and hand-sewn masks.
In Italy, an engineering startup called Isinnova 3D printed venturi valves (which connect oxygen masks to respirators) that were needed to help keep patients alive. Italy’s hospitals were quickly overrun by COVID-19 patients, leading to a critical shortage of respirators and ventilators. Isinnova founder Christian Fracassi and his team reverse engineered the valve, and printed 100 of them in one day.
Ventilator manufacturer Medtronic has also shared all of its design files so that other entities can print parts for its equipment without having to reverse-engineer or redesign the parts.
3D printer companies and engineering software providers are also contributing resources. For example, Siemens has made its Additive Manufacturing (AM) Network (along with its 3D printers) available to the global medical community to speed design and production of medical components. The AM Network connects users, designers and 3D-print service providers to enable faster production of spare parts for machines like ventilators. Siemens’ designers and engineers are a part of the AM Network, and can answer design requests and help convert designs into printable files. Afterwards, these components can be printed via medically certified 3D printers of partner companies that are also part of the AM Network.
3D Systems, Stratasys, HP and other 3D printer companies have organized initiatives to create PPE, asking for contributions from engineers and providing links to downloadable designs and other information.
“There is enormous potential and value that the additive industry can provide at this time of crisis,” says Radhika Krishnan, executive vice president and general manager, software, at 3D Systems.
Engineers Called on to Contribute
More help is needed, and there are a variety of ways that engineers can get directly involved. In the case of the CoVent-19 Challenge, a group of Boston-based anesthesiology residents have put out a call to the engineering community to participate in an eight-week hackathon hosted on the engineering collaboration portal GrabCAD.com to design a rapidly deployable ventilator for patients with COVID-19-related lung problems.
The CoVent-19 Challenge is open to teams and individuals anywhere. Finalists will work directly with Stratasys 3D printing experts and the CoVent-19 Challenge team to produce prototypes for physical testing. Just 24 hour after the initiative was announced on April 1, it had already received two entries as CAD assembly files. As of this writing, there are now 32 entries.
According to the dedicated portal for the event, called INNOVATE2VENTILATE, “The deficit of ventilators in the United States alone is expected to be in the hundreds of thousands. Our goal is to rapidly expand the capacity of hospitals, by expanding our current ventilation abilities, in order to survive the peak of this pandemic.”
Up to $10,000 in prizes are available for the challenge.
To participate, visit www.coventchallenge.com.
A brand new organization, Open Source Medical Supplies, has also been created to provide rapid, open-source COVID-19 response around the globe. The gruop has been designated as a non-rofit, and has partnered with RESOLVE.ngo, Schmidt Futures, and Toyota Research Institute to bring together maker, hospital systems, manfuacturers and other groups to fill gaps in the medical supply chain. You can learn on the group's Facebook page.
America Makes
Youngstown, Ohio-based America Makes (the National Additive Manufacturing Innovation Institute) is collaborating with the U.S. Food and Drug Administration (FDA), the Department of Veterans’ Affairs, and the National Institutes of Health (NIH), to better coordinate industry response to COVID-19 supply chain issues.
As part of that effort, the organization has set up the COVID-19 Health Care Needs and Additive Manufacturing Capabilities Repository, where additive manufacturers can upload 3D print designs to be reviewed and placed on the NIH 3D Print Exchange.
You can find more information at americamakes.us/covid-19.

Printed in Italy on a ProX 6100 in Medical Grade Nylon. Image courtesy of Christian Fracassi and team, Isinnova; 3D Systems.
Regulatory Compliance Still Required
While the availability of additional PPE and ventilators is important, the equipment still needs to meet specific requirements in order to be safe and effective in the field. The FDA has provided some guidance in this area. The NIH also provides designs that have been reviewed for clinical use.
3D Systems has also established a Materials Technology Overview Chart for medical equipment that engineers and makers can refer to when working on these projects, along with a face shield frame design on its COVID-19 page.
“We’ve seen concerns expressed regarding medical devices being created using materials that are not biocompatible,” the company said in a statement. “For all medical devices, 3D Systems complies with guidance from the FDA – testing the safety and efficacy of all devices. Recently, the FDA has issued several emergency use authorizations for things such as PPE for healthcare providers that are allowing companies to get clearance more quickly than previously possible. Additionally, FDA, VA, America Makes & CDC have all published guidance and 3D Systems is collaborating with all these organizations in our response efforts.”
“It’s critically important that you are following the regulatory guidelines and doing this the right way,” Krishnan says. “The additive manufacturing industry at large is not completely clued into the nuances of what it takes to build parts for healthcare applications, so that is one thign we have tried to be very mindful about. The good news is that there are a lot of tools that can get the job done.”
Subscribe to our FREE magazine, FREE email newsletters or both!
Latest News